Partnership Opportunities
Unlocking Success: Explore Exclusive Event Partnership Opportunities
With ongoing investments in Treg-directed therapies, the excitement surrounding these immune re-balancing approaches is reaching new heights. As we eagerly await the release of clinical data to validate their effectiveness, there's no better time to present your solutions to a highly engaged community of Treg leaders.
Partnering with the Treg-Directed Therapies Summit is not just a collaboration; it's a strategic investment in propelling Treg therapeutic development forward. This event stands out with its unwavering focus on Treg Therapies, providing an exclusive platform to engage with visionaries at the forefront of autoimmunity treatments.
Why Partner With Us in 2026?
Establish Your Industry Leadership:
Position yourself as a recognized authority in the Treg space at the premier event dedicated to transformative Treg innovations. Join the top minds and companies driving progress, showcasing your expertise and reinforcing your status as a trusted industry leader
Elevate Your Brand Presence:
Boost your brand’s visibility and influence, before, during, and beyond the summit. Use our platform to create a memorable impact that resonates with your target audience
Network with Key Decision-Makers:
Connect with an exclusive network of C-suite executives, VPs, and other senior leaders in discovery, R&D and clinical development. These decision-makers are actively seeking impactful strategies to advance clinical translation, providing an ideal setting for meaningful collaboration and idea exchange
Gain Exclusive Market Insights:
Stay ahead with insider knowledge on the latest advancements and strategies in Treg therapeutics. Learn from leading biotech and pharma companies about emerging trends, breakthrough research, and future plans to maintain a competitive edge in this dynamic field
Hear from Previous Attendees:
“Excellently organized with great speakers on critical topics.”
Amy Rosenberg, Senior Director, Immunology & Consultant, EpiVax, 2024 Attendee
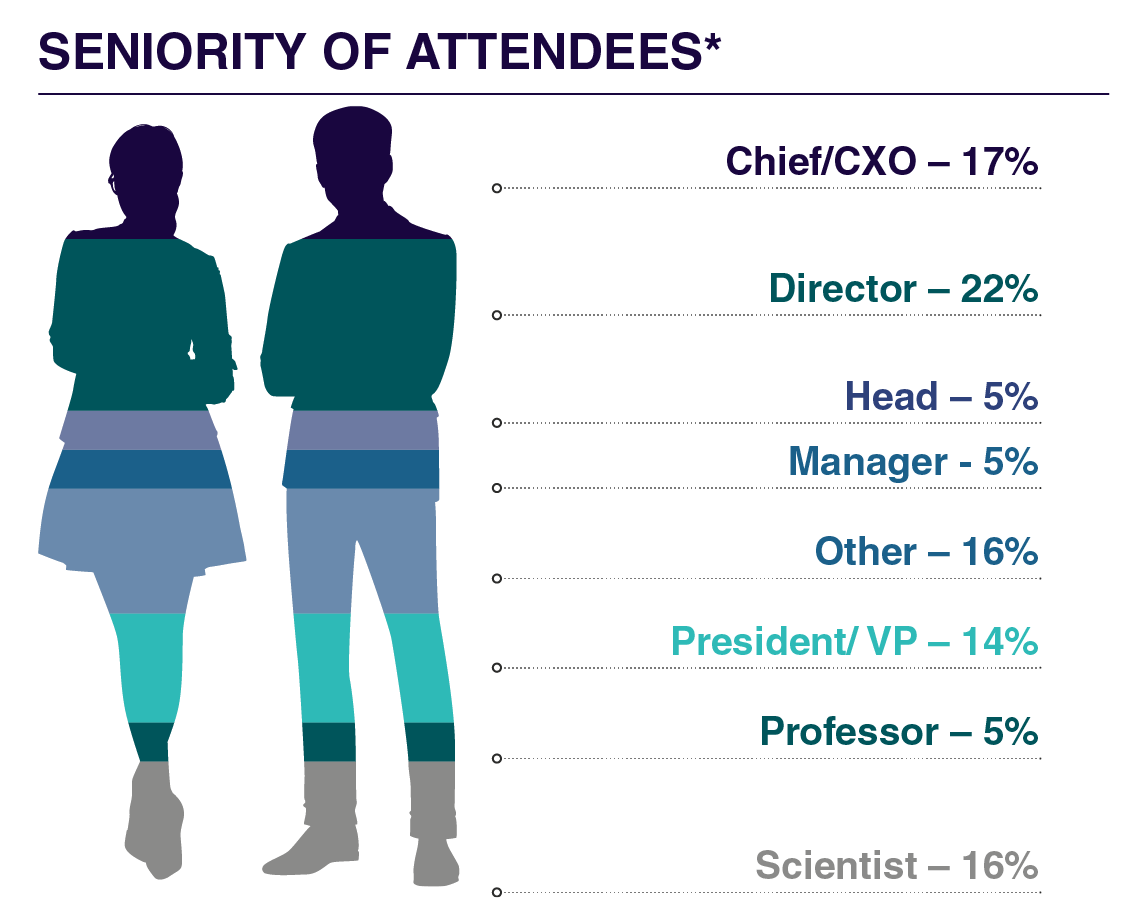
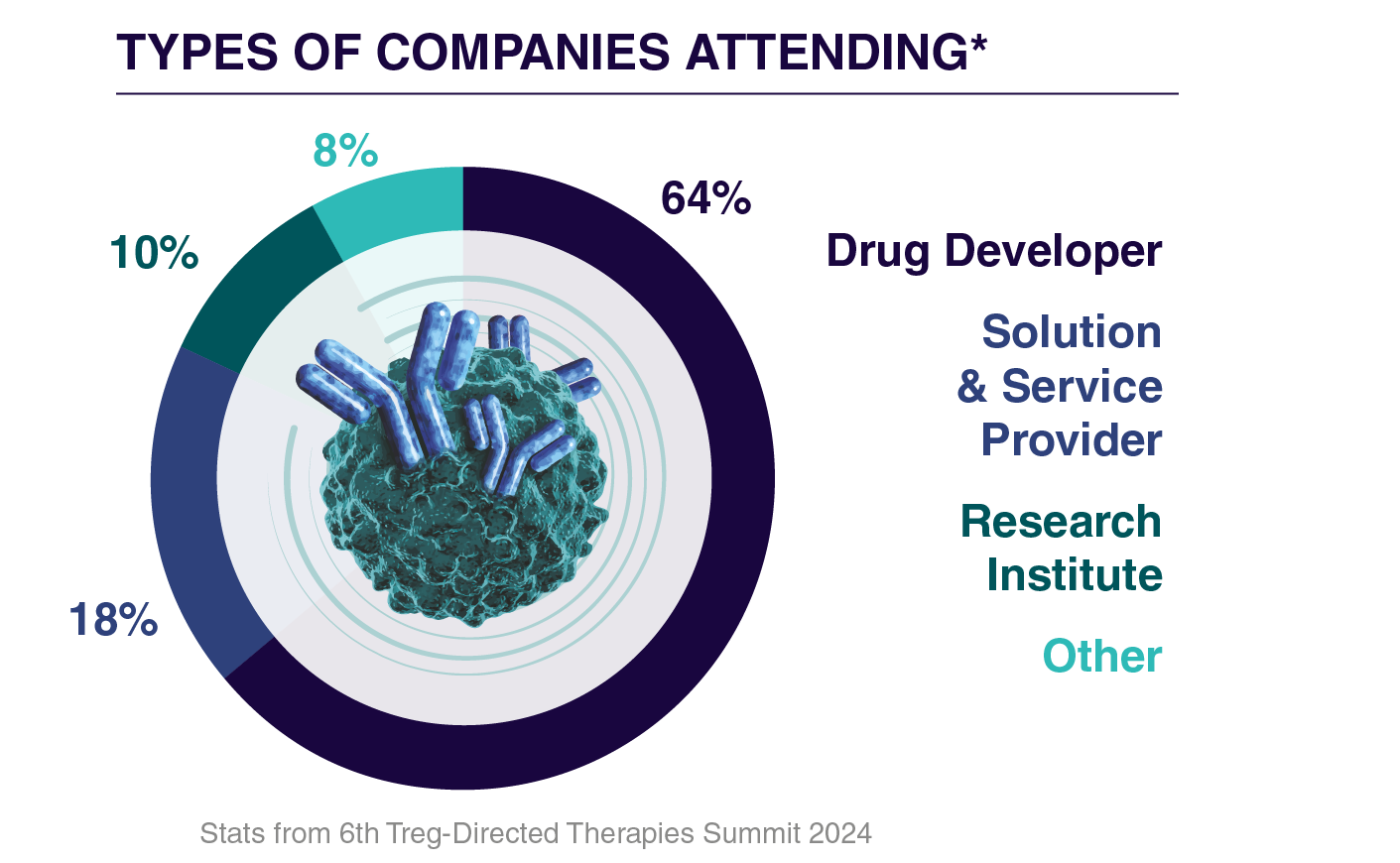
Who Will You Meet?
Explore Related Events
Introducing the Cell Therapies Event Series, a collection of 26 established events curated to enable this community to achieve their clinical and commercial potential.
Having trouble downloading the brochure? Let us know here and we'll email it to you instead.
